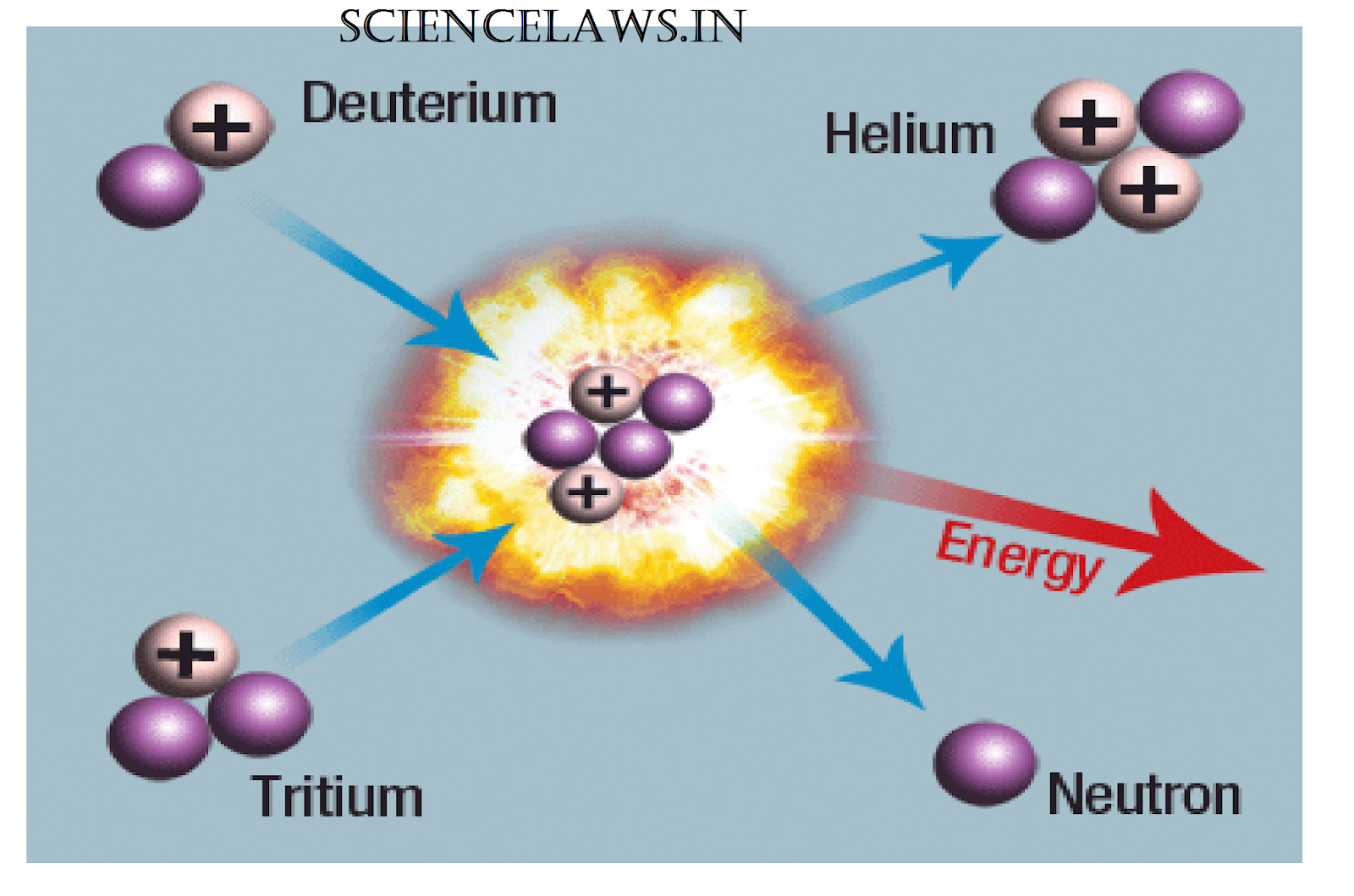
The U-236 nucleus then rapidly breaks apart into two smaller nuclei (in this case, Ba-141 and Kr-92) along with several neutrons (usually two or three), and releases a very large amount of energy.Īmong the products of Meitner, Hahn, and Strassman’s fission reaction were barium, krypton, lanthanum, and cerium, all of which have nuclei that are more stable than uranium-235. This inherent safety feature can be overwhelmed in extreme circumstances.\): When a slow neutron hits a fissionable U-235 nucleus, it is absorbed and forms an unstable U-236 nucleus. In case the reactor overheats and boils the water away, the chain reaction terminates, because water is needed to thermalize the neutrons. High energy is needed to fuse two or more atoms together. Comparatively, less energy is needed to split an atom in a fission reaction. Fusion reactions occur in stars and the sun. Fission reactions do not occur in nature naturally. Control rods adjust neutron flux so that criticality is obtained, but not exceeded. The energy released during nuclear fusion is several times greater than the energy released during nuclear fission. A pressurized water reactor is cleverly designed to control the fission of large amounts of 235U, while using the heat produced in the fission reaction to create steam for generating electrical energy. Figure 4 shows a schematic of a reactor design, called the pressurized water reactor. Water is very effective, since neutrons collide with protons in water molecules and lose energy. As noted in Chapter 32.5 Fusion, energy is released if the products of a nuclear reaction have a greater binding energy per nucleon ( \boldsymbol, it is thus necessary to slow down (“thermalize”) the neutrons. Five nuclear equations and a graph are shown. (b) The larger fission products of U-235 are typically one isotope with a mass number around 85105, and another isotope with a mass number that is about 50 larger, that is, about 130150. (credit: Kalmthouts)įission is the opposite of fusion and releases energy only when heavy nuclei are split. Figure 21.4.3: (a) Nuclear fission of U-235 produces a range of fission products. 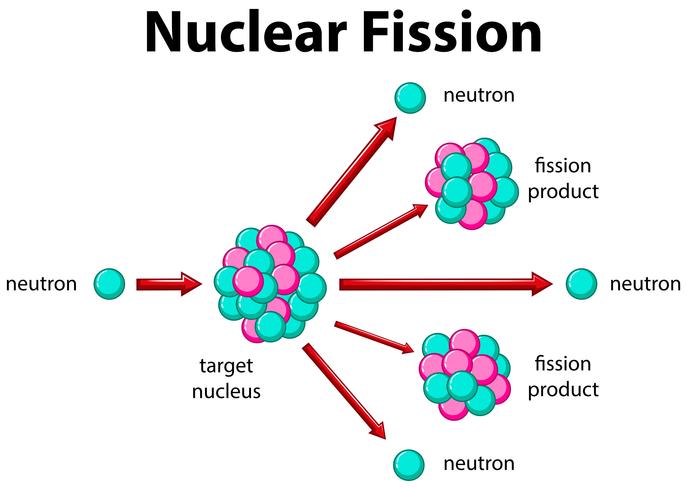
The reactor is in the small domed building to the left of the towers. 
The cooling towers are the most prominent features but are not unique to nuclear power. About 16% of the world’s electrical power is generated by controlled nuclear fission in such plants. The people living near this nuclear power plant have no measurable exposure to radiation that is traceable to the plant. China is building nuclear power plants at the rate of one start every month. France provides over 75% of its electricity with nuclear power, while the US has 104 operating reactors providing 20% of its electricity. By the end of 2009, there were 442 reactors operating in 30 countries, providing 15% of the world’s electricity. 
Whereas nuclear power was of little interest for decades following TMI and Chernobyl (and now Fukushima Daiichi), growing concerns over global warming has brought nuclear power back on the table as a viable energy alternative. Hundreds of nuclear fission power plants around the world attest to the fact that controlled fission is practical and, at least in the short term, economical, as seen in Figure 1. Controlled fission is a reality, whereas controlled fusion is a hope for the future. This type of reaction is called nuclear fission. As an example, let us have a look at how uranium-235 fissions. (Figure 1.6.10 for an example of uranium-235 fission fragment spectrum). Nuclear fission is a reaction in which a nucleus is split (or fissured). Nuclei show various types of reaction: For example, one nuclide splits into two or more fragments. Two types of fission reactions are possible: spontaneous fission and induced fission.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
